Quick note before we begin: I’m not a medical doctor, and nothing here is medical advice. This is for educational purposes only. Talk to an actual doctor before doing anything with your health. Cool? Cool.
Unless you’ve been living under a rock for the last few years, you’ve probably heard of these new weight loss drugs everyone’s talking about.
Celebrities are taking it, friends in your group chat are sharing their opinions, your aunt’s Facebook page is full of conspiracy theories about them, and your annoying colleague Bob lost a bunch of weight using them and never shuts the fuck about it – so what’s the deal?
Let’s talk about that.
What are GLP-1 weight loss drugs?
Ozempic. Wegovy. Mounjaro.
If you haven’t heard at least one of these names in the last year or two, I’m genuinely impressed by your ability to avoid the internet.
The names mentioned above are the brand names. The drugs behind them are semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro), and they mostly fall under the same umbrella: GLP-1 receptor agonists.
(Tirzepatide is technically a dual GLP-1/GIP agonist, meaning it targets a second hormone receptor too — but for the purposes of this article, I’m grouping them together because the mechanisms and outcomes are similar enough. But also because typing “glucagon-like peptide-1” dozens of times makes me want to smash my head through my laptop, repeatedly, and I’d rather not start the weekend with a concussion. Anyway, back to the topic.)
GLP-1 is a hormone your body naturally produces in the gut after you eat that signals to your brain that you’re full, slows down how quickly food leaves your stomach, and triggers insulin release to manage blood sugar. The GLP-1 receptor agonists are drugs that mimic this hormone, but they’re way more potent, which means their effects last longer; the net result is that you feel fuller for longer, have fewer food cravings, and eat less.
As such, GLP-1s aren’t “fat burners” – i.e. they don’t directly reduce body fat. Rather, they reduce appetite, allowing people to sustain a calorie deficit, which, when done consistently over time, can lead to weight loss.
Do they actually work?
You bet they do.
A 2025 joint advisory from the American College of Lifestyle Medicine, the American Society for Nutrition, the Obesity Medicine Association, and The Obesity Society reviewed the evidence and found that GLP-1s produce average weight losses of 15–21% in the major clinical trials, though in real-world use, where adherence is lower and conditions less controlled, that typically drops to around 8–11%.
For obese individuals — the population these drugs were designed for — those losses come with meaningful improvements across a wide range of conditions, such as cardiometabolic risks/events, heart failure, obstructive sleep apnea, prediabetes, and more. None of this should be surprising, since the health benefits of losing excess weight are well-documented.
Notably, most of the weight loss occurs in the first year of treatment, with results plateauing by around 16–18 months. There isn’t that much data beyond two years, so what happens after that point if someone continues using them isn’t well established.
This sounds too good to be true–what’s the catch?
As they say, there’s no free lunch, and GLP-1s do come with some side effects.
Most of these seem to impact the gastrointestinal (GI) system. Per the same review mentioned earlier, nausea affects roughly 25–44% of users; diarrhoea affects 19–30%; vomiting shows up in 8–24% of cases; and abdominal pain affects about 9–20%.
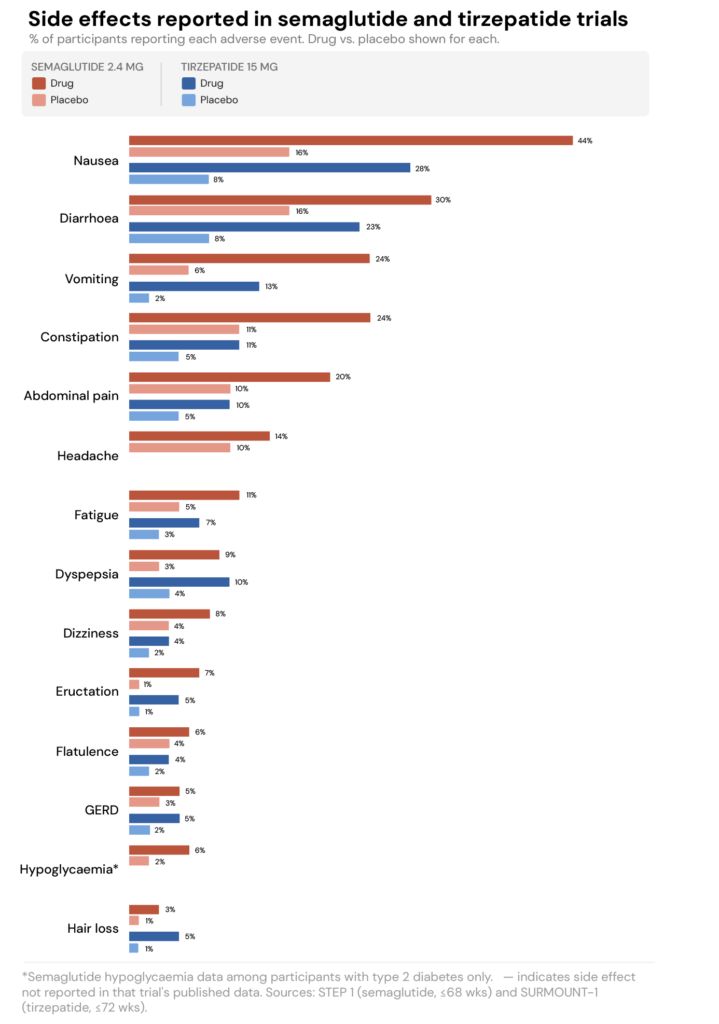
The good news (?) is that these side effects tend to be worse when you first start, or when the dosage is increased, and they often improve as your body adjusts.
A bigger concern, though, is what happens to body composition during GLP-1 use.
It’s not just fat loss
Earlier in the article, I noted that GLP-1s significantly reduce appetite, leading to a similarly significant reduction in caloric intake. According to the review by Mozaffarian and colleagues, reductions in caloric intake can range from 16% to 39%.
To contextualise that: the average adult eating around 2,000 calories a day would, at the lower end, drop to roughly 1,600 calories per day. At the upper end, they’d be eating around 1,200 calories per day. For one person, that might result in an appropriate intake level (e.g., a small, sedentary female), but for another, it could be a very aggressive deficit.
And when you lose weight fast, it’s not just fat you’re losing–you also risk losing muscle.
For example, a network meta-analysis of 22 randomised controlled trials found that lean tissue accounted for roughly 25% of total weight loss on GLP-1s on average. Mozaffarian et al. drew from modelling data and found that, in the absence of resistance training, muscle accounts for approximately 10–15% of total weight lost in women and 20–25% in men.
It’s worth highlighting that the muscle loss seen with GLP-1s isn’t dissimilar from the muscle loss you see during weight loss without these drugs.
So, it’s not that these drugs are suddenly eating up people’s muscles–they tank your appetite, which can lead to a significant caloric deficit. If this isn’t compensated for with enough protein and resistance training, then, yes, muscle loss will occur.
There’s also a less-discussed micronutrient angle worth noting. People with obesity already tend to under-consume certain vitamins and minerals, compared to the general population, and that’s before any deliberate calorie restriction. Once you throw in aggressive calorie reduction, the risk of falling short on essential nutrients increases, which is another reason to be intentional about food quality.
What happens when you stop?
The research here isn’t encouraging.
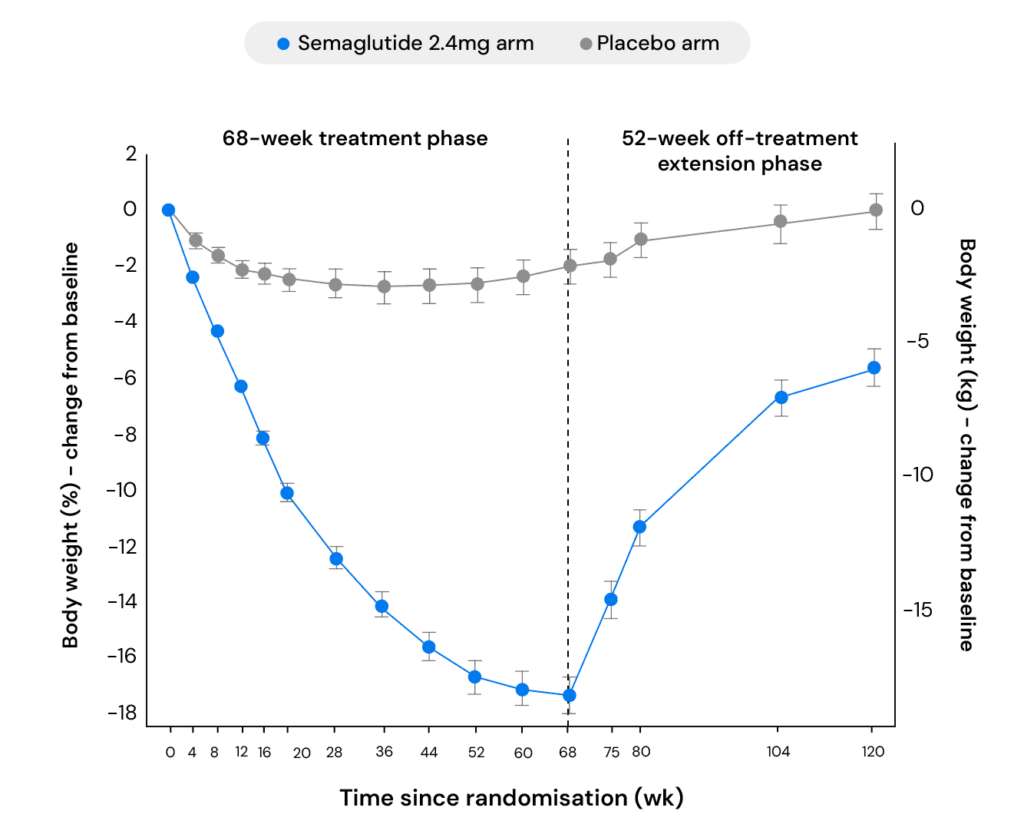
Wilding and colleagues followed participants through 68 weeks of treatment—during which the semaglutide group lost an average of 17.3% of body weight—and then for a further year after treatment was discontinued. In that time, participants regained roughly two-thirds of the weight they’d lost.
A separate trial compared what happened when participants continued using semaglutide versus switching to placebo, while both groups received identical lifestyle interventions. Those who switched to a placebo gained an average of 6.9% of body weight over the following 48 weeks, with more than 80% regaining weight.
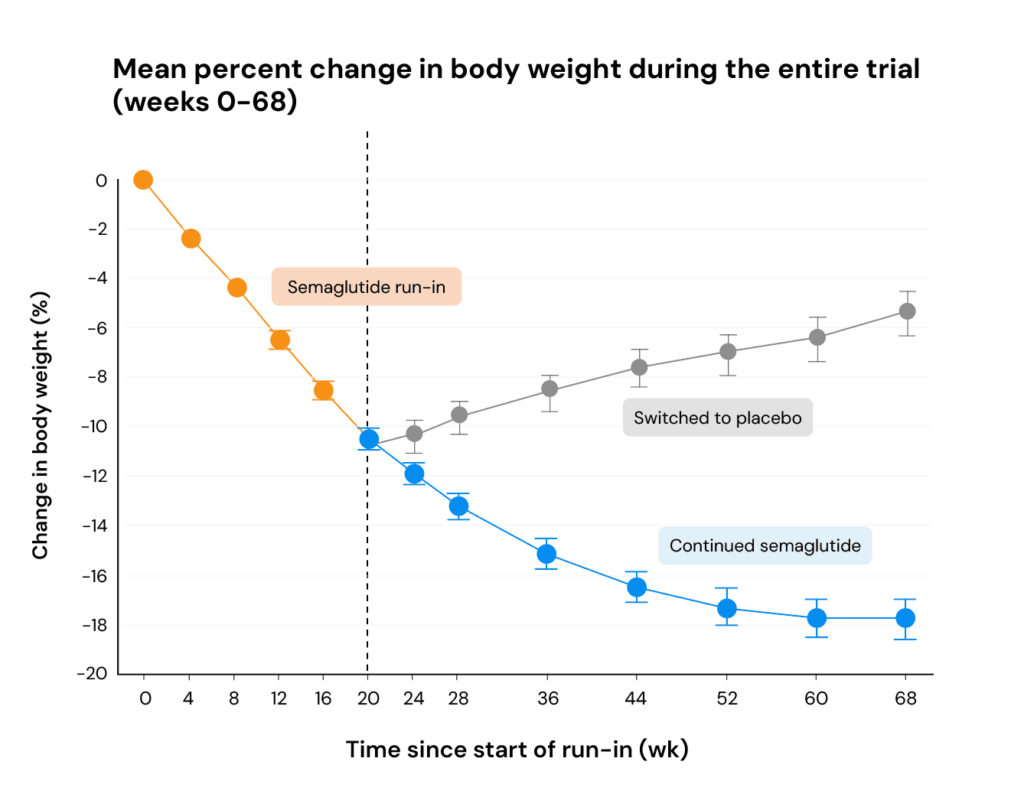
For many people, it seems GLP-1s may need to be used indefinitely to maintain the benefits, much like you’d need to continue taking blood pressure medication to maintain blood pressure control.
Bottom line
GLP-1s are genuinely effective for weight loss, providing real health benefits for people with obesity-related health conditions. There can be side effects (primarily GI-related), and muscle loss is a real concern. However, the side effects seem short-lived, and any muscle loss can be mitigated with resistance training and adequate protein intake.
The harder reality is that most people will likely need to use these drugs indefinitely to maintain the benefits. But that’s also a reason to focus on the healthy habits that support your health and body composition, drug or no drug.
As effective as GLP-1s may be, they’re still a tool that needs to be combined with broader lifestyle changes, such as eating a nutrient-rich diet with enough protein and staying physically active, with a focus on resistance training.
Yes, they make behaviour change easier, but individuals taking them still need to change their behaviour–there’s no drug (yet) that can get you to do the thing. That’s still on you.
Thanks for reading. If you enjoyed this, you’d love the Vitamin
95% of my new content is only sent to my email list. One email every Thursday, filled with actionable, evidence-based fitness advice to help you with your goals. If you enjoyed this, you’ll love my emails. You can learn more and subscribe for free here.